
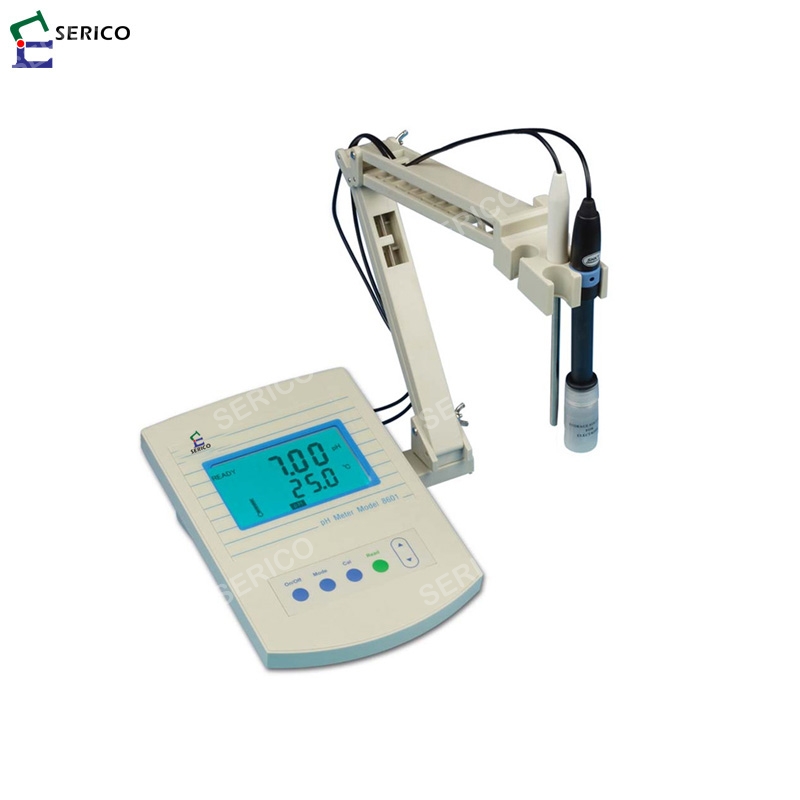
Hydration is required for the ion exchange PH electrode, due to the nature of its construction, needs toīe kept moist at all times.
#PH METER FREE#
Such as CCI4, which does not have any free hydrogen ions. They are not designed to be used in solvents, PH electrodes are available in a variety of styles for both laboratoryĪnd industrial applications. That it be interfaced only with equipment with high impedance The voltage signal produced by the pH probe In summary, the measuring pH electrode deliversĪ varying voltage and the reference electrode delivers a constant The potential of the reference portion is produced by the internalĮlement in contact with the reference fill solution. Junction site of the measuring portion is due to the free hydrogen One portion is called the measuring pH electrode, the The most widely used variety, there are actually two electrodes The alkali metal ions of the glassĪnd the hydrogen ions in solution undergo an ion exchange reaction, This glass is typicallyĬomposed of alkali metal ions. In some cases, voltages can also be read for special applications or for use with ion-selective or Oxidation-ReductionĮlectrodes are constructed from a special composition glass which Impedance amplifier that accurately measures the minute electrode voltages and displays the results directly in pH units on either an analog The input impedance of the pH meter and leakage resistances are therefore important factors. The pH electrode has very high internal resistance, making the voltage change with pH difficult to

The reference electrode output does not vary With the changes in the relative hydrogen ion concentration inside and outside of the bulb. The pH probe is a hydrogen ion sensitive glass bulb, with a millivolt output that varies The pH electrode can be thought of as a battery, with a voltage that varies with A pH tester system consists of three parts: a pH probe,Ī reference pH electrode, and a high input impedance meter. More accurate pH measurements are obtained with a digital pH meter. These indicators have limitations on their accuracy, and can be difficult to interpret correctly in colored or murky samples. The relationship between hydrogen ions and hydroxyl ions in a given solution is constant for a given set of conditions, either one can beĪ rough indication of pH can be obtained using pH papers or indicators, which change color as the pH level varies. If equal amounts of H+ and OH- ions are present, the material is neutral, with a pH of 7.Īcids and bases have free hydrogen and hydroxyl ions, respectively.If the OH- concentration is greater than H+, the material is basic,.Is greater than OH-, the material is acidic i.e., the pH measurement is less than 7. The quantitative information provided by the pH tester expresses the degree of the activity of an acid or base in terms of hydrogen ion activity. The pH value of a substance is directly related to the ratio of the hydrogen ion and the hydroxyl ion concentrations.
